How Do You Know if a Complex Ion Is Inert or Labile
2.eleven: Ligand Lability
- Page ID
- 195679
Lability refers to the ease with which ligands are replaced in coordination complexes. Scandium is referred to as "labile" in the following example.
[Sc(OH2)six]Clthree + 6 NaSCN —> Naiii[Sc(SCN)half dozen] + iii NaCl (very fast!)
Lability refers to how easily metal-ligand bonds are broken. A compound in which metal-ligand bonds are hands broken is referred to as "labile". A compound in which metallic-ligand bonds are more difficult to break is referred to as "inert".
Henry Taube (Nobel Prize, 1983) tried to understand lability past comparing the factors that govern bond strengths in ionic complexes to observations virtually the rates of reaction of coordination complexes. He saw some things that were unsurprising. He likewise drew some new conclusions based on ligand field theory.
Exercise \(\PageIndex{ane}\)
In which compound from each pair would you await the strongest ionic bonds? Why?
a) LiF vs KBr
b) CaCl2 vs. KCl
- Answer
- Answer a
-
The ions in LiF are both smaller than in KBr, then the force of attraction between the ions in LiF is greater because of the smaller separation betwixt the charges.
- Respond b
-
Calcium has a 2+ charge in CaClii, whereas potassium has only a + accuse, so the chloride ions are more than strongly attracted to the calcium than to the potassium.
Taube observed that many Thousand+i ions (Yard = metallic) are more labile than many M+3 ions, in general. That isn't too surprising, since metallic ions function as electrophiles or Lewis acids and ligands function as nucleophiles or Lewis bases in forming coordination complexes. In other words, metals with college charges ought to exist stronger Lewis acids, so they should bind ligands more tightly.
All the same, there were exceptions to that general rule. For example, Taube as well observed that Mo(V) compounds are more labile than Mo(Iii) compounds. That means there is more going on hither than simply accuse effects.
Another cistron that governs ionic bail strengths is the size of the ion. Typically, ions with smaller atomic radii form stronger bonds than ions with larger radii. Taube observed that Al3+, V3+, Feiii+ and Ga3+ ions are all about the same size. All these ions commutation ligands at nigh the aforementioned rate. That isn't surprising, because they have the same charge and the same radius.
Still, Crthree+ is also well-nigh the aforementioned size as those ions and it also has the aforementioned charge, but it is much less labile. One time again, there are exceptions to our regular expectations based on unproblematic electrostatic considerations.
Furthermore, second- and third-row transition metals (Y-Cd and Air-conditioning-Hg) are much more inert than first-row transition metals (Sc-Zn). That is a footling surprising, since those lower metals are much larger than the starting time row metals.
However, it gives us a clue about other factors that are playing a role in lability. In ligand field theory, second- and tertiary-row metals have much larger d orbital splitting energies than do beginning-row metals. That is sometimes explained in terms of lengthened orbitals on these larger atoms forming stronger bonds to ligands, and reminds us that we are not just dealing with electrostatic interactions.
Taube wondered whether d electron configuration influenced whether a compound is labile or inert. That idea forms the footing of Taube's rules about lability.
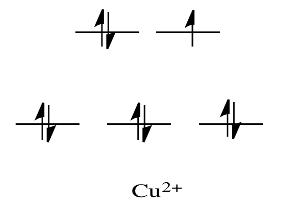
For case, metals like Ni2+ and
Cu2+ are very labile. The d orbital splitting diagrams for those compounds would accept d electrons in the eg gear up. Call back, the eg set arises from interaction with the ligand donor orbitals; this set corresponds to a σ antibonding level.
By comparison, V2+ is rather inert. The d orbital splitting diagram in this example has electrons in the t2g set, but none in the eastwardg set.
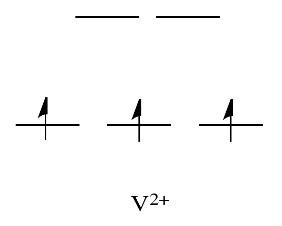
And so, having electrons in the higher energy, antibonding due eastg level weakens the bail to the ligand, and then the ligand tin be replaced more easily. In the absence of those college free energy electrons, the bond to the ligand is stronger, and the ligand isn't replaced as easily.
On the other paw, metals like Caii+, Sc3+ and Ti4+ are pretty labile. The d orbital splitting diagrams in those cases are pretty simple: there are no d electrons at all in these ions.
That ways having no electrons in these mostly non-bonding levels leaves the complex susceptible to ligand replacement. But it's hard to see why population of an orbital that is mostly not-bonding would have an effect on ligand bond strength.
Instead, this factor probably has something to do with the part of ligand substitution that nosotros have ignored so far. Not merely does one ligand need to leave, but a 2d i needs to bail in its place. Then, having an empty orbital for the ligand to donate electrons into (or, put another fashion, non having electrons in the way that may complicate donation from the ligand) makes that office of the reaction easier.
Practice \(\PageIndex{two}\)
Some metals, like Mn2+, tin can be either labile or inert, depending on whether they are high spin or depression spin. Explain why using d orbital splitting diagrams.
- Answer
-
Add texts hither. Do not delete this text showtime.
Practice \(\PageIndex{3}\)
Predict whether the following metals, in octahedral complexes, are labile or not.
a) Co3+ (high spin)
b) Coiii+ (low spin)
c) Ironii+ (low spin)
d) Atomic number 262+ (high spin)
east) Zn2+
- Answer
- Answer a
-
labile (electrons in higher free energy d orbital fix)
- Answer b
-
not labile (all electrons in lower free energy d orbitals)
- Respond c
-
not labile (all electrons in lower free energy d orbitals)
- Reply d
-
labile (electrons in higher free energy d orbital set)
- Answer e
-
labile (electrons in college energy d orbital ready)
pollardprionsprould1946.blogspot.com
Source: https://chem.libretexts.org/Bookshelves/General_Chemistry/Book%3A_Structure_and_Reactivity_in_Organic_Biological_and_Inorganic_Chemistry_(Schaller)/III%3A_Reactivity_in_Organic_Biological_and_Inorganic_Chemistry_1/02%3A_Ligand_Binding_in_Coordination_Complexes_and_Organometallic_Compounds/2.11%3A_Ligand_Lability#:~:text=A%20compound%20in%20which%20metal,referred%20to%20as%20%22inert%22.

0 Response to "How Do You Know if a Complex Ion Is Inert or Labile"
Post a Comment